This post was written by James Edlund, MD, medical director, PPG – Wound Care, Parkview Health.
The use of hyperbaric air—ambient or room air at increased atmospheric pressure—has been proposed and practiced since before the discovery of oxygen. Hyperbaric oxygen therapy (HBOT) today involves controlled conditions requiring a licensed medical provider's prescription and oversight. However, in recent years, alternative versions, known by names such as "HBOT Lite", "Hyperbaric Lite" and "Mild Hyperbaric," have emerged in non-clinical environments marketed for off-label therapies as well as sold for in-home use. To help our community better understand the difference between true HBOT and HBOT Lite, this post aims to clarify how each practice operates and the associated risks of non-indicated use.
What is "true" hyperbaric oxygen therapy?
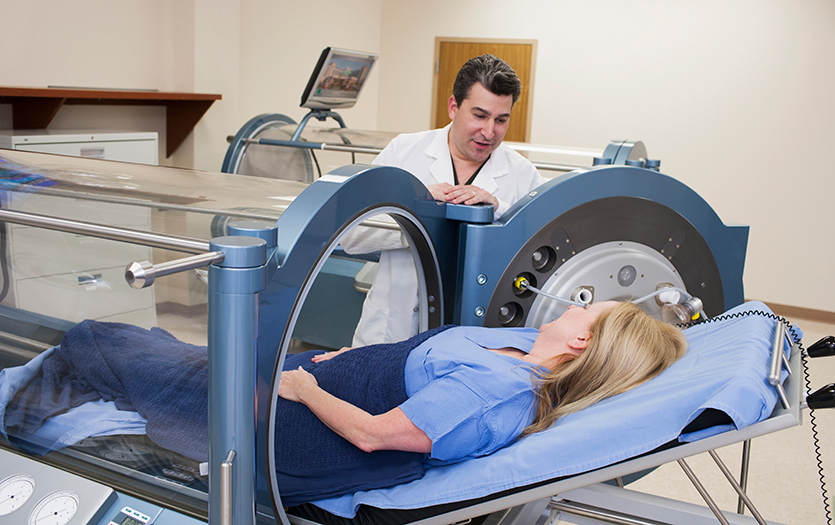
HBOT is a noninvasive medical treatment that uses pure oxygen in a pressurized environment to address various conditions. The Parkview Center for Wound Healing offers this service to provide advanced therapy for patients with wounds that aren't responding to traditional treatment.
Medical professionals perform the therapy within a hard-sided hyperbaric chamber that meets the standards set by the American Society of Mechanical Engineers (ASME) for Pressure Vessels for Human Occupancy (PVHO) and the National Fire Protection Association code (NFPA 99). These governing bodies have approved hard-sided chambers to tolerate pressure up to three atmospheres.
During HBOT, the patient's entire body is placed in the chamber and treated with medical-grade oxygen (>99%) at a pressure of no less than 2.0 ATA for 90 to 120 minutes. Under these conditions, oxygen acts as a regulated drug overseen by the U.S. Food & Drug Administration (FDA).
What is HBOT Lite?
Conversely, "HBOT Lite" involves exposure to oxygen at lower pressures and concentrations. Unlike true HBOT, when performed in non-clinical settings, this practice frequently uses soft-sided, portable or inflatable chambers that seal with a zipper. The FDA categorizes these as Class II Medical Devices and has approved them solely for the purpose of treating Acute Mountain Sickness. These chambers only tolerate a pressure of 1.4 atmospheres and are not approved for supplemental oxygen.
Are there any risks associated with using HBOT Lite?
The Undersea and Hyperbaric Medicine Society (UHMS) helps define the safe practice of hyperbaric medicine. It is an international organization founded in 1967 that oversees diving medicine and clinical applications of HBOT.
When administered appropriately, inflatable and portable pressure chambers present no risk. However, when applied in circumstances outside its intended purpose, they may prove hazardous due to the following complications:
Inadequate pressure. Exposure to treatment pressures less than 1.4 ATA while breathing air does not meet the definition of therapeutic HBOT and does not achieve the minimum pressure and oxygen levels required for any UHMS and FDA-approved indication. Patients utilizing mild or lite HBOT may risk relying on treatments that provide no real benefit without proper pressure and oxygen levels.
Misuse of supplemental oxygen. To reiterate, milder chambers are not currently designed or approved to safely contain supplemental oxygen. However, in numerous instances, commercial businesses and consumers have purchased fabric or soft-sided chambers and modified them for use with supplemental oxygen.
Repurposing these chambers is especially dangerous as the materials involved may risk tearing or leaking oxygen into the ambient air. Although oxygen itself does not burn, it significantly increases the flammability of other materials, causing them to burn faster and hotter.
When performed outside of the supervision of trained experts and clinical settings certified to prevent and respond to such a threat, this procedure can create dangerous circumstances for all involved. These risks become especially severe when performed in a residential setting, where appropriate safeguards are not in place.
Furthermore, when administered as a supplement, the FDA considers medical-grade oxygen a drug that requires a prescription to treat an eligible medical condition and supervision from a healthcare professional throughout its use. HBOT may involve multiple sessions over the course of days or weeks. Without oversight, prolonged exposure to high oxygen levels can become toxic to the brain and lungs.
Delayed proper treatment. In response to the increased incidence of HBOT off-label use, the FDA released a consumer update letter titled Hyperbaric Oxygen Therapy: Get the Facts. In this publication, the FDA warns against the promotion of HBOT Lite and similarly named services for unapproved conditions like autism or cancer.
Advertisements citing this developing research can inadvertently mislead patients into believing milder therapies are adequate for their health concerns. Opting for these services due to ambiguous marketing tactics may cause patients to delay the pursuit of medically proven interventions. This postponement can worsen their health outcomes as many chronic and progressive conditions require timely and targeted treatments.
What types of conditions is HBOT used to treat?
While there are ongoing clinical trials aimed at exploring HBOT's efficacy for treating other conditions at various pressures, at this time, they are not FDA-approved indications for HBOT. The currently accepted indications for HBOT are as follows:
-
Air or gas embolism
-
Arterial insufficiencies
-
Central retinal artery occlusion
-
-
Carbon monoxide poisoning
-
Clostridial myonecrosis
-
Compromised grafts of flaps
-
Acute traumatic ischemia
-
Decompression sickness
-
Delayed radiation injuries
-
Soft tissue radio necrosis
-
Osteoradionecrosis
-
-
Sudden sensorineural hearing loss
-
Intracranial abscess
-
Necrotizing soft tissue infections
-
Refractory osteomyelitis
-
Severe anemia
-
Severe thermal burns
-
Avascular necrosis (aseptic osteonecrosis)
Final thoughts
At the Parkview Center for Wound Healing, our team frequently provides HBOT to treat conditions like diabetic foot ulcers and delayed radiation injuries. While we specialize in these conditions, that is not to exclude many other indications, our team of experts is equipped to address conditions that we are not consulted for.
Due to the cost of services associated with HBOT, we thoughtfully assess each case to determine whether this therapy is the best option for the patient. We aim to ensure our patients are aware of and have access to all viable treatment options. Although we may not approve every HBOT request, we are always willing to consider consulting for most conditions on the list.
If your healthcare provider recommends HBOT, we strongly advise that you pursue treatment at a hospital or facility that has been inspected and is properly accredited by the UHMS. Parkview Centers for Wound Healing offers HBO as adjunctive therapy, which does not require a physician referral. If you are dealing with a chronic wound or need additional information and support, contact any of our three locations for help.



